PersonaCRC assesses the risk of spread, recurrence, and overall survival, to help clinicians identify the most effective treatment strategies and improve patient outcomes.
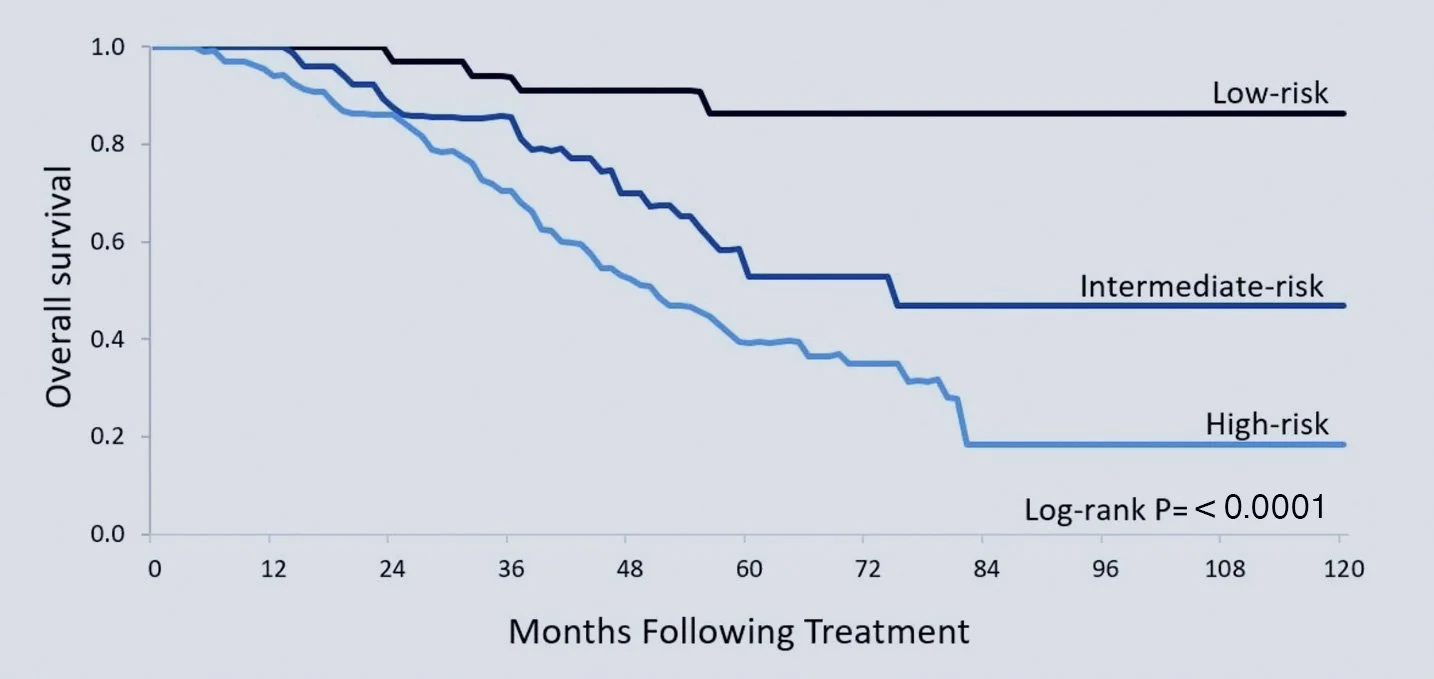
Metastasis accounts for the majority of cancer-related deaths, and treatment decisions are often made without clear prognostic tools. PersonaCRC provides evidence-based stratification to support more precise decision-making. By integrating RNA sequencing data with clinical insights, our platform stratifies metastatic colorectal cancer into low, intermediate, and high-risk groups with over 90% accuracy.
This clarity empowers clinicians to:
Identify which patients are most likely to benefit from curative local therapy versus those who require systemic treatment.
Avoid unnecessary or ineffective systemic treatments
Provide patients with evidence-based guidance at a critical decision point
PersonaCRC Key Outcomes:
PersonaCRC is supported by decades of research and independently validated with Phase 3 New EOPC trial data.
>90% accuracy in classifying colorectal liver metastases
50% difference in 5-year survival between low- and high-risk groups
Identification of a low-risk subgroup with markedly improved survival and potential for cure
Is PersonaCRC right for your patient?
PersonaCRC is ideal for patients who are:
Diagnosed with colorectal cancer with metastasis to the liver
Undergoing or considering treatment options
In need of precise risk stratification
Seeking personalized therapy
The PersonaCRC test is not recommended for:
Patients with primary colorectal cancer only
Patients with metastasis beyond the liver

Why PersonaCRC changes everything.
Actionable Reporting
Clear risk tiers and therapy guidance, with results delivered within 48 hours of sequencing data.
Validated Biomarker Technology
Validated performance: Independent validation with Phase 3 trail data, peer-reviewed, with over 90% accuracy.
Impactful Patient Outcomes
Avoids unnecessary treatments, and reduces costs.
Enhancing Treatment Decisions with PersonaCRC
PersonaCRC provides integrated prognostic risk classification to personalize treatment decisions— helping clinicians precisely define disease biology, manage patients with confidence, and identify those with potential for cure.
Ordering Process:
Physician Orders Test
The treating physician orders the PersonaCRC test for their patient with the test request form (TRF)
Click to View TRF >Tissue Sample Sent
The liver metastasis tissue is sent to Caris Life Sciences for RNA sequencing. Sequencing data are then securely transferred via AWS to Protean BioDiagnostics. No additional procedures are required from the patient.
AI Analysis
PersonaDx applies its proprietary AI model to generate a molecular subtype and integrated risk group.
Results & Treatment Plan
A Protean BioDiagnostics board-certified pathologist reviews the analysis and delivers the final clinician-ready report to guide treatment planning.
"We provide a unique platform and robust research pipeline that brings unparalleled precision to oncology, potentially reducing morbidity, mortality, and unnecessary patient suffering at a lower cost of care."
— Dr. Ralph Weischelbaum, National Academy of Medicine Member

Ready to transform patient care?
Join leading oncologists who are already using PersonaCRC to deliver precision medicine.
Contact Us:
Phone: 1 (754) 242 9682 | Email: info@proteanbiodx.com

Publications, Research, and White Papers
Listed below are some of the latest research and data highlighting the real-world impact of PersonaCRC on metastatic cancer prognosis and treatment planning. Access the in-depth white papers via the publication links for more detailed insights.
PersonaDx, Inc. | N/A | PersonaDx, Inc. | Data on File | Internal PersonaDx data on file. PersonaDx, Inc., [Chicago, IL].
World Health Organization | 2022 | World Health Organization | Cancer: Key Facts | World Health Organization. (2022). Cancer: Key facts. https://www.who.int/news-room/fact-sheets/detail/cancer
JAMA Oncology | 2023 | Katipally, R. R., et al. | Integrated Clinical-Molecular Classification of Colorectal Liver Metastases: A Biomarker Analysis of the Phase 3 New EPOC Randomized Clinical Trial | Katipally, R. R., et al. (2023). Integrated clinical–molecular classification of colorectal liver metastases: A biomarker analysis of the Phase 3 New EPOC randomized clinical trial. JAMA Oncology, 9(9), 1245–1254. https://doi.org/10.1001/jamaoncol.2023.2535
Pitroda, S. P., et al. (2018). Integrated molecular subtyping defines a curable oligometastatic state in colorectal liver metastasis. Nature Communications, 9, 1793. https://doi.org/10.1038/s41467-018-04278-6
Lancet Oncology | 2020 | Bridgewater, J., Pugh, C., Maishman, T., et al. | Systemic chemotherapy with or without cetuximab in patients with resectable colorectal liver metastasis (New EPOC): long-term results of a multicentre, randomised, controlled, phase 3 trial | Bridgewater, J., Pugh, C., Maishman, T., et al. (2020). Lancet Oncology, 21(3), 398–411. https://doi.org/10.1016/S1470-2045(19)30798-3
Morris, V. K., Kennedy, E. B., Baxter, N. N., et al. (2023). Treatment of metastatic colorectal cancer: ASCO guideline. Journal of Clinical Oncology, 41(2), 678–702. https://doi.org/10.1200/JCO.22.01690